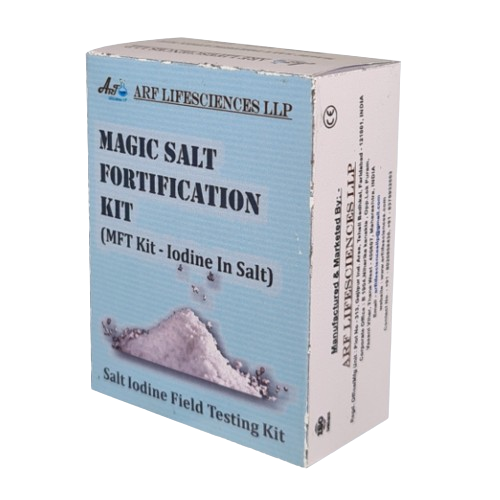
Description
Benefits
- Low Cost Per Test
- Portable
- Comprehensive Kit
- Unique
- Accurate
Precautions & Expiry
- This kit can be stored at ambient temperature and relative
- humidity (20-90%) in any part of the country in sealed conditions.
Keep away from direct sunlight - 6 Months Shelf life









Reviews
There are no reviews yet.